Why might my patient’s fasting bile acid concentration be higher than the postprandial bile acid concentration?
Bile Acid Basics:
Measurement of fasting and postprandial bile acids is a relatively sensitive and easily performed screening test to detect hepatobiliary disease or portosystemic shunts (PSS). Normally, after a meal is ingested, the gallbladder releases bile acids into the intestines. Later, bile acids are absorbed into the portal circulation and most are removed by the liver. Increased bile acids in the systemic circulation occur if there is decreased removal from portal circulation (e.g., hepatic failure, PSS) or decreased biliary excretion (e.g., hepatic or post-hepatic cholestasis).
Case Example:
A 5-year-old neutered male dog had a nine-month history of seizures which were increasing in frequency. Physical examination was unremarkable. Serum biochemical and CBC results were within reference limits, except for the following:
-
Bile Acids
Patient
Ref. Int.
-
Fasting34.4 μmol/L0.5-8.7
-
Postprandial23.0 μmol/L0.5-23.4
This pattern of bile acid results is common. Review of 470 canine bile acid profiles submitted to DCPAH from July 15, 2009 through July 14, 2010 revealed that 109 (23%) had higher fasting than postprandial bile acid concentrations, and 76 of these profiles had values within reference limits or only mildly increased. This pattern may occur when: 1) gallbladder contraction is stimulated by something other than ingestion of food, 2) the dog ingests something unbeknownst to the owner or veterinarian, or 3) sample tubes are mislabeled. The fasting value may exceed the upper reference limit for the postprandial sample because of variations in gallbladder contraction (e.g., the fasting value does not reflect baseline concentration) and in time from contraction to sampling.
Interpretation of bile acid results can be challenging and should include consideration of history, physical examination, and other diagnostic findings (e.g., serum biochemical profile, imaging of the liver). Values that are only mildly increased, as in this dog, may or may not be caused by hepatobiliary disorders or PSS. This dog had no other findings to support hepatobiliary disease or PSS (baseline plasma ammonium concentration and hepatic ultrasound were within normal limits). The seizures were felt to be nonhepatic in origin and the dog responded to anticonvulsant drugs.
Why should I send slides along with fluid for cytologic examination? Is there an additional charge?
Fluid Submission Basics:
It is best to submit slides made immediately after fluid collection (see Fluid Cytology Preparations from the Fall 2008 issue) because cells deteriorate during transit to the laboratory, particularly if fluids lack sufficient protein to stabilize cell membranes (e.g., washes, cerebrospinal fluid). Cellular deterioration makes it difficult to assess cell details and even to determine cell types. Moreover, contaminating bacteria may proliferate and be phagocytized in vitro during transit, making it difficult or impossible to differentiate sepsis from contamination.
When smears are submitted with fluid, the submitted smears and smears made in our laboratory from the fluid are examined. There is no additional charge to review the submitted smears.
Case Example:
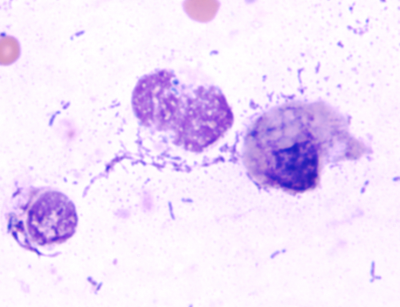
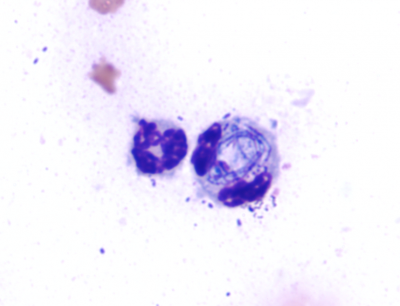
The client submitted direct smears, concentrated smears made from sediment that was resuspended after centrifugation of an aliquot of the wash fluid, and tracheal wash fluid from a two-month-old male horse that was dyspneic and febrile. Auscultation revealed wheezes and crackles in all lung fields. The samples arrived two days after collection. Concentrated smears made at DCPAH from the submitted fluid contained large numbers of lysed and deteriorated nucleated cells (Figure 1). Most could not be identified as to type. There were many extracellular bacteria. Intracellular bacteria were not confirmed. In contrast, the submitted concentrated smears had numerous neutrophils (Figure 2), many containing intracellular bacteria. Based upon the submitted smears, a diagnosis of septic neutrophilic inflammation was possible.